
Food for Your Brain
April 25, 2021
Foods That Boost Your Immune System
May 16, 2021Low Quality Hydrogen Tablet Reviews and Test Results

Often, I allude to the “old” hydrogen tablets, which I did not manufacture, that utilize different technology. These are the hydrogen tablets that launched before I started my R&D process, by “MK” who I mention in the “Hydrogen Tablets: Origins” article.
These archaic tablets were never filed with the U.S. FDA, so they do not have New Dietary Ingredient Status, as the tablets I developed do. These tablets have not gone through any clinical research, as contrasted by the many clinical trials and case studies the open cup hydrogen tablets I developed have been used in. These “other hydrogen tablets” do not have any posted third-party testing results, or accreditation, such as the gas chromatography results we posted or our International Hydrogen Standards Association (IHSA) certification.
Additionally, these tablets, to the best of my knowledge, did not use an appropriate grade of magnesium. This is evidenced by the testing I commissioned on the three separate brands utilizing this technology all coming back with 3–4 times higher lead content, despite our tablets utilizing 40–50% more magnesium. I want to make it clear that they are not in violation of California Proposition 65, even with these elevated levels. With these tablets, you can take about six of them a day before the State of California, which has the strictest laws regarding contaminants in the USA, deems it a potential hazard. For my tablets, you can take 20+ per day before reaching the threshold. Efforts need to be made to ensure compliance with California Proposition 65. One high-profile example was when two large, respected U.S. –based manufacturers (Nutraceutical Corp and Graceleigh) were sued by the California attorney general for high levels of lead in their baby formula.
You can see a redacted version of a certificate of analysis (CoA) on one of our production runs below, and the CoAs of each of the three brands of the “old” hydrogen tablets I analyzed in their respective write-ups. Also attached is a CoA of our magnesium results, which show about 90% of magnesium in ionic form in the water (some will complex into salts such as magnesium malate), followed by 100% delivered as free magnesium ions by the time it hits your stomach. This percentage is much, much lower for the “old” hydrogen tablet technology, as shown in the CoAs below in each respective segment.
Finally, these tablets should be expired. They are many years old. They are still being sold. See the videos below.
CoA From Natural Wellness Now Open Cup Hydrogen Tablets
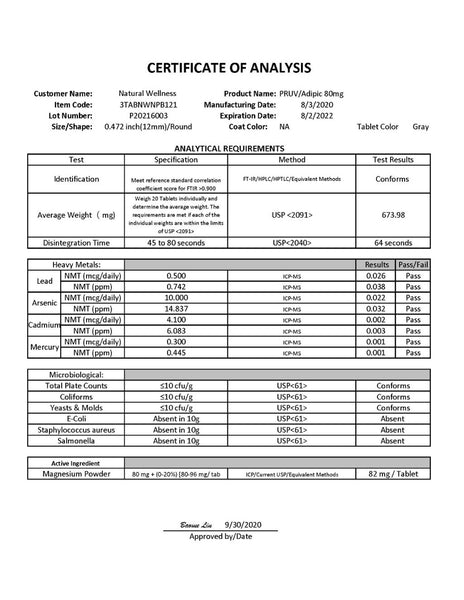
Magnesium Test Results

AlkaViva Hydrogen Tablets
AlkaViva were the very first tablets to hit the market. They are the “oval”-shaped tablets I spoke about in “Hydrogen Tablets: Origins” part 1, where MK purportedly made them in his kitchen, supplying to JH. As you can see from the video below, the tablets were manufactured in October 2014, 6.5 years ago, and are still being sold! In the United States, supplements are supposed to have a 2-year shelf life, unless stability data justifies otherwise. These tablets are 4.5 years beyond this window, and still being sold.
I tested the tablets at 0.5–0.7ppm (parts per million) in 500ml in an open cup, and also in a sealed bottle following the directions on the packaging. See the video below for my testing. This is roughly what I tested them at 6+ years ago, so the stability has remained good. The issue with the long shelf life is the plastic packaging, which can start to degrade over time and contaminate the product.
Video Results
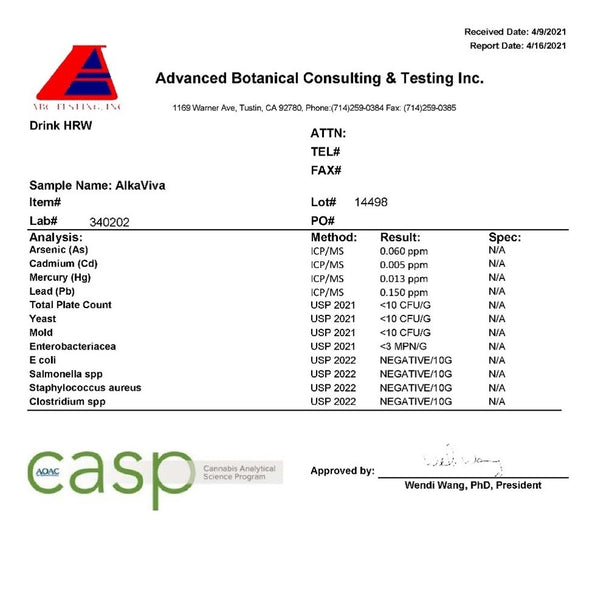
Interestingly, the lab reported lower hydrogen levels than I did; finding between 0.2–0.3ppm in 500ml of water. If we go by the lab results, the AlkaViva hydrogen tablets fall below what the IHSA determines to be the minimum therapeutic concentration. Additionally, only 50% of the magnesium is detectable in the water, meaning the majority is either unreacted or has complexed. This demonstrates the inefficiency of the tablets, and potential hazards.
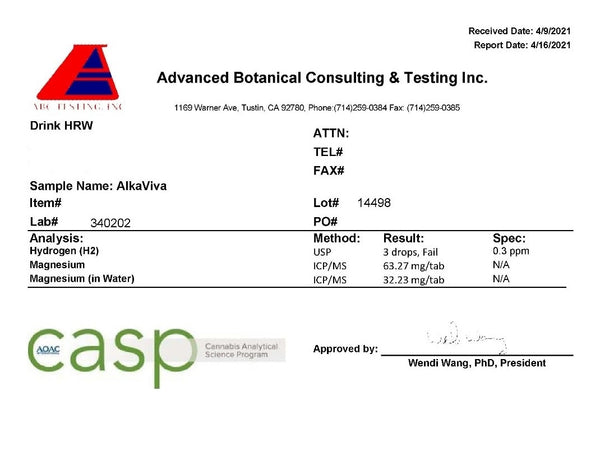
Fortunately, it seems there has been no microbiological growth after this extended time.
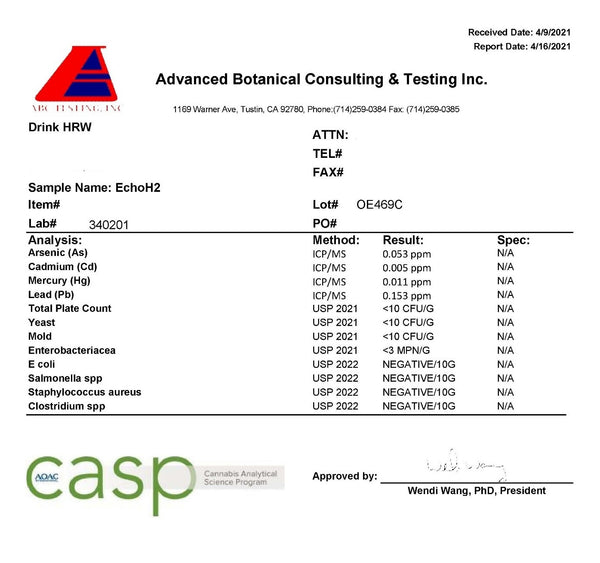
Echo Hydrogen Tablets
Echo was one of the first brands of tablets to hit the market. Like AlkaViva, it appears its first, and likely only, batch of product is still in circulation. The “manufactured on” date shows May 2015, so the tablets are 6 years old — 4 years past the typical expiry date, when stability data is not present. I haven’t had many dealing with Echo over the years, nor have I tried to pursue their business. There are a few reasons for this:
- If it’s stilling selling a batch from 6 years ago, then its volumes aren’t great.
- It associates with many distributors and influencers I want absolutely no connection to, such as Mike Adams, Ty Bollinger, etc.
- The things I have heard reported from distributors of Echo regarding its claims make my jaw drop. Claims such as they “place their machines and the hydrogen tablets next to special crystals during storage, which imparts quantum energy into the products and makes them superior to competitors.” I will emphasize that I have never heard this directly from Echo H2, however, enough others have told me this that I’m forced to consider the fact it is likely true. That is, likely true that they are saying this, NOT true that they are actually imparting some sort of magical quantum energy into their products.
The Echo tablet took forever to dissolve. At the 5:00 minute mark you can see that water splashed on a tablet, and the surface did not react. I could speculate on the issues with formulation and manufacturing, as I have seen this before, but I won’t. The result is that the Echo H2 hydrogen tablets take over 6 minutes to dissolve in an open cup in room temperature water. The Echo H2 tablets measured 0.3–0.5ppm in the open cup and 0.3–0.5ppm in a sealed bottle in my testing:
The lab results showed 0.4ppm, as it failed on the fifth drop. Additionally, the lab results show that the tablets are 10% below the magnesium content claimed on the label, and less than half of what was found, and only 40% of what is claimed, can be detected after disintegration in the water. I want to point out that this level of hydrogen concentration falls below what the IHSA has determined is the minimum therapeutic concentration.
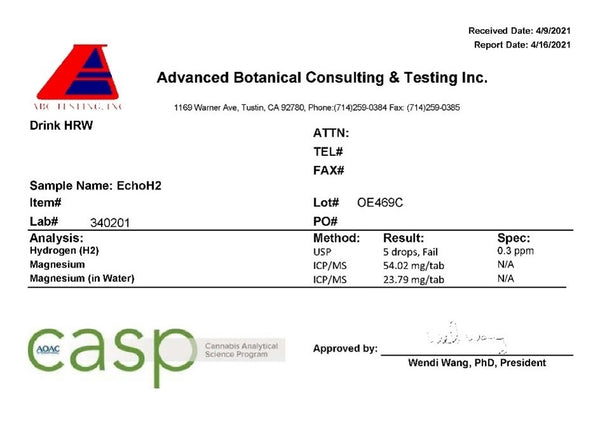
Like with the AlkaViva tablets, there is fortunately no microbiological growths or issues.
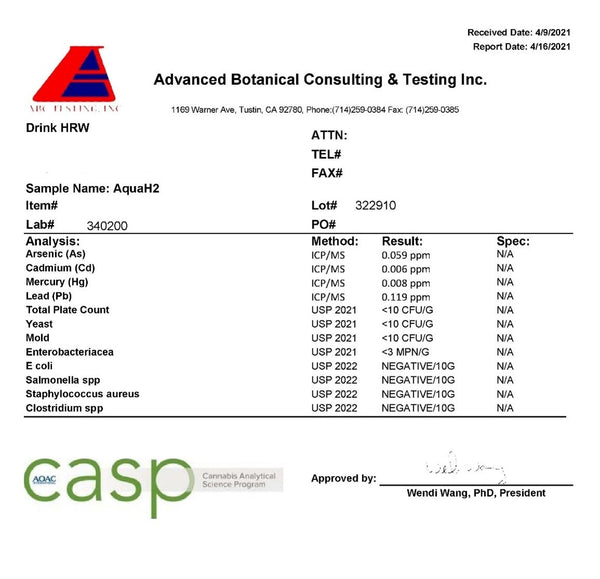
Aqua H2
Aqua H2 is the brand owned and operated by MK, the inventor and manufacturer of the hydrogen tablets utilized by AlkaViva and Echo. There is some controversy regarding the stated expiration date on the Aqua H2 tablets, as there are numerous complaints on the Aqua H2 Amazon page regarding fake expiration stickers placed on top of the actual expiration dates on the outer containers, with the proper expiration dates stamped onto the card.
https://www.amazon.com/Molecular-Hydrogen-Tablets-Water-Aquah2/dp/B01E9NVYNU/ref=sr_1_1?dchild=1&keywords=aqua+h2&qid=1619195053&sr=8-1
The product I received did not have this phenomenon; although, it is entirely possible MK could have packaged new goods. Also, it is legal to put an expiry date based on packaging date, not manufacture date, so long as analysis is done after packaging. I have it on good authority, from many individuals, that MK was caught off guard by my company’s takeover of the industry, being stuck with millions of hydrogen tablets with no buyers for his technology. There was a mass exodus from MK as a manufacturer to me, with virtually every brand I was willing to work with switching their products over to my technology.
The expiration date on the box that Aqua H2 comes in now states a date of Dec. 31/2023. My suspicion is that he either packaged the remainder of his tablets that were manufactured years ago with a fresh expiration date, or he manufactured new tablets — but not by legal means considering laws surrounding elemental magnesium or perhaps even cGMP requirements. I know for a fact that the manufacturing facility he had set up to run his tablets stopped manufacturing tablets; its press was sold in the summer of 2018.
Additionally, I heard from several parties that MK tried to sell customized tablets manufactured in non-cGMP conditions after 2018. And I have it on good authority that he is currently living in India.
The likelihood that MK has found a new cGMP facility in the USA, during the pandemic, to run a new lot manufacturing with an explosive ingredient (during manufacturing) is extremely unlikely, especially since many manufacturers are dropping smaller clients right now due to high demand. If MK did find a new facility under these conditions, it seems astronomically unlikely he invested in the proper safety measures to manufacture these tablets legally and safely. Actually, I can write about it now upon reviewing the NDA I signed with his facility, which is now expired. The facility he used previously had not employed proper safety measures and seemed completely unaware of the laws governing the manufacture of these goods when I toured the facility. It seems unlikely that he would be compliant with the law now, with no appreciable demand or business, when he wasn’t when he had demand.
As for the testing results from these tablets, in an open cup I measured somewhere between 0.7–0.9ppm — a higher reading than either AlkaViva or Echo H2. Just as in the other tablets, the reading in a sealed bottle was identical, also measuring 0.7–0.9ppm when prepared as instructed.
Before scratching your head regarding the third-party lab results and the large disparity between my results, watch the video starting at 8:15 (above). A high amount of grey scum was present on the surface of the water, which I scooped off. This scum is grey as it contains unreacted elemental magnesium. This magnesium, if poured into the beaker, would directly react with the reagent, mimicking how hydrogen turns the blue drops clear. This grey scum may explain the high reading from the third-party lab report, which was much higher than mine, at 1.6ppm. Another explanation could be improper mixing. I have heard from brands that used to demo MK’s later generation tablets that they were largely inconsistent. One tablet would be impressive, and then the next would give a low reading. Again, this is just speculation. What we do know is that the measured magnesium content is almost 15% below what is claimed on the label, and the magnesium measured in the water after disintegration is only at around 50% of what is claimed on the label. Here are the lab reports:

Like with the AlkaViva and Echo H2 hydrogen tablets, there is fortunately no microbiological growths or issues.
Conclusion
Not all hydrogen tablets are created equally. I have written about many “hydrogen scams”, and will write about more. While these tablets are not “scams”, they deliver only a fraction — 3-5% to be precise — of the hydrogen concentration the hydrogen tablets I developed deliver. They come with higher levels of lead, and the conditions they were made in are questionable. As always, “Buyer beware”.










1 Comments
What a helpful analysis and explanation of the wide range of concentration…and possible scams. Thank you! I continuously recommend DrinkHRW to my clients because it’s the only brand I can trust.