Science Behind Muscle Fatigue and How We Can Prevent It
December 4, 2021
Significance of Telomere Length For Aging and Human Health
January 30, 2022Sleep – Universal Function But Unclear Origins

This is the first of a 10-part series exploring sleep, its regulation, sleep loss, and its consequences, parasomnias, sleep optimization, and much more.
Everyone sleeps, and each of us has some certainty about “how much we need”. Indeed, most people sense that sleep is essential, but few appreciate how much depends on this immensely complicated behavior. From insulin sensitivity to vigilance, from concussion recovery to dreaming, the quality and duration of your sleep will impact nearly every aspect of your life. To illustrate its importance, consider the following: (1) total sleep deprivation in rats leads to death, but without a clear anatomical cause [1]; (2) a single night of sleep loss in humans can negatively impact endocrine, inflammatory, and metabolic function [2]; (3) the behavior of sleep emerged and has persisted across almost all species [3], despite conferring no obvious ecological advantage. Think about this last point outside of the context of society: sleep makes us vulnerable to predation and restricts us from feeding and procreating – it runs opposite to the very bedrock of our survival, yet sleep can be observed in just about every species of animal and insect. To quote Allan Rechtschaffen, a pioneer in sleep research: “If sleep does not serve an absolutely vital function, it is the greatest mistake evolution ever made.”
Jellyfish sleep [4], as do platypus [5], fruit flies [6, 7], and zebrafish [8]. A phylogenetic examination of these species’ shared ancestors suggests that sleep is an ancient behavior and/or that the need for it is near-universal. So, what universal advantage or protection might sleep provide species with such biological and ecological diversity? While truly novel hypotheses emerge every-so-often, the majority of sleep scientists seem to fall in the following camps: (1) that sleep is essential for energy conservation or restoration [9]; (2) that sleep emerged for the strengthening, maintenance and/or weakening or neural associations (and thus, for memory) [10-12]; or, more recently, (3) that sleep permits “waste clearance” in the brain, much like the peripheral lymphatic system [13, 14]. While there is no agreed-upon fundamental function of sleep, we now know a great deal about how it is regulated and about what we stand to lose when sleep is acutely or chronically disrupted.
The Two Process Model
The “Two Process Model”, originally described by Alexander Borbély, is a widely accepted framework for understanding the regulation of sleep. It posits that the circadian rhythm (Process C) and the sleep homeostat (Process S) are distinct processes which interact to predict sleep-wake behavior [15].
Process C – The Circadian Rhythm
Just about every cell in your body has a built-in clock that keeps track of time in a circadian manner (“circadian” is Latin for “about a day”). This pace-keeping machinery is a complex transcription-translation feedback network that reliably oscillates at a rhythm of about a day, driving cell- and organ-specific processes and, more broadly, human behavior [16]. It is important to note that each of these clocks keeps track of time on their own (even if you take them out of the body [17]) and they rely on external signaling factors to coordinate their activity.
The most important non-visual function of the eyes is to entrain your circadian rhythm. When light enters the eye, specialized photoreceptors called “opsins” transduce this light into an electrical signal. This signal is then projected via the retinohypothalamic tract to the suprachiasmatic nucleus (SCN) of the hypothalamus, where it sets the time of our “master clock” [18, 19]. The master clock in our SCN is a tiny population of neurons (about 20,000 in mice) with the important task of generating an alerting signal with daily variation and is essential for influencing the timing and duration of sleep [20]. This molecular machinery is also responsible for telling the rest of the body “what time it is”. Without the coordinating influence of the SCN, sleep and activity become arrhythmic and there are severe disturbances to physiological processes [20-22].
The timing of our sleep is set in part by our circadian rhythm, and our circadian rhythm is principally influenced by the timing of our light exposure. Circadian phase-shifting occurs when light in your environment is inconsistent with the “expectations” of your internal clock – like using your cell phone in bed, suffering through shift work, or travelling to a new time zone. In addition to light, the timing of food [23], exercise [24], and even socialization [25] can influence circadian rhythms. Consistent sleep timing is associated with robust hormonal and metabolic health, improved sleep, and improved energetics [26-29].
Process S – The Sleep Homeostat
Process S interprets waking experience and produces pressure to sleep (aka “sleep pressure”, “sleep drive”, or “sleep propensity”) [30]. If you stay awake long enough, you will eventually get tired; and, once you are rested, additional sleep is difficult to come by. People tend to take these phenomena for granted, perhaps because these are some of our first (and certainly most consistent) experiences from birth. This active effort to balance sleep and wake can be observed across all sleeping species – in fact, it is a defining feature that helps to separate sleep from other quiescent states, like coma. “Homeostasis” is the active defense of a setpoint. Just like the thermostat in your home, the job of the Sleep Homeostat – or “Process S” – is to defend a setpoint (temperature) by exerting an appropriate pressure (heating or cooling). We can measure changes to sleep pressure by several behavioral metrics (like sleep latency and sleep duration), and through EEG by measuring the synchronization of neurons in the cortex [15]. I’ll speak at length about how we measure sleep using polysomnography and the importance of distinct neural oscillations in a later blog.
While we know a great deal about circadian rhythms, relatively little is known of the systems underlying Process S. How does the brain interpret waking experience? How does sleep resolve the feeling of sleepiness? In sheep, the central administration of cerebrospinal fluid (CSF) taken from sleepy animals is sufficient to increase sleep pressure in well-rested animals [31]. Several studies suggest that some sleep-promoting factor (maybe adenosine) accumulates during wake (gradually increasing sleep pressure) and then breaks down during sleep [32]. There is also evidence that sleep is a necessary follow-up to the metabolically demanding and complex processes associated with neuroplasticity [12] – more on this later.
Summary
Sleep is a highly conserved behavior across species, regulated by two separate but related processes. At the molecular level, the circadian rhythm (Process S) is a transcription-translation feedback loop responsible for entraining our biology and behavior to a daily changing environment. A central “master clock” called the suprachiasmatic nucleus, is responsible for generating an alerting signal for promoting wakefulness, as well as coordinating the activity of the molecular clocks that exist throughout the brain and body. Our sleep homeostat (Process S) regulates the accumulation of sleep pressure as we stay awake and the subsequent dissipation of sleep pressure across consolidated sleep.
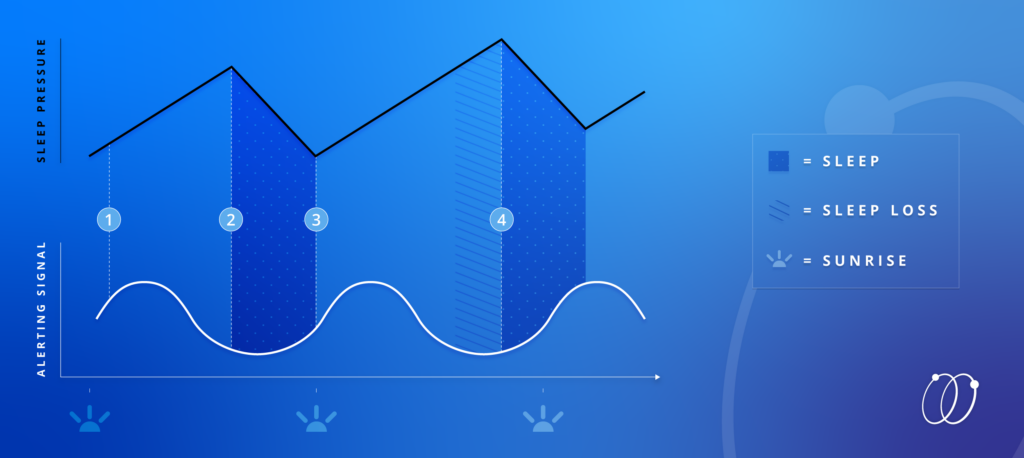
An adaptation of Borbély’s Two-Process Model (above) illustrates the interactions of these dynamic systems:
(1) During the day, the alerting signal (white) from Process C is high, and the pressure to sleep (black) increases as we stay awake.
(2) Once sleep pressure is high enough, and the alerting signal is low, we are naturally driven to fall asleep. As we sleep, our sleep pressure decreases.
(3) Once the alerting signal is high, and sleepiness is low, we are naturally driven to wake up.
(4) If we stay awake longer than normal, our internal pressure to sleep will continue to increase until we satisfy our need for rest. When we finally do sleep, our sleep is likely to intrude into the next day’s normal waking period. When this happens repeatedly, we can shift the rhythm of our circadian clock.
In the next installation, we’ll discuss the diverse biological and psychological consequences of sleep loss for the individual and the broad societal cost, as well as what happens when your brain and body must adapt to less sleep.
Works Cited
- Everson, C. A., Bergmann, B. M., & Rechtschaffen, A. (1989). Sleep deprivation in the rat: III. Total sleep deprivation. Sleep, 12(1), 13-21.
- Mullington, J. M., Haack, M., Toth, M., Serrador, J. M., & Meier-Ewert, H. K. (2009). Cardiovascular, inflammatory, and metabolic consequences of sleep deprivation. Progress in cardiovascular diseases, 51(4), 294-302.
- Keene, A. C., & Duboue, E. R. (2018). The origins and evolution of sleep. Journal of Experimental Biology, 221(11), jeb159533.
- Nath, R. D., Bedbrook, C. N., Abrams, M. J., Basinger, T., Bois, J. S., Prober, D. A., … & Goentoro, L. (2017). The jellyfish Cassiopea exhibits a sleep-like state. Current Biology, 27(19), 2984-2990.
- Siegel, J. M., Manger, P. R., Nienhuis, R., Fahringer, H. M., Shalita, T., & Pettigrew, J. D. (1999). Sleep in the platypus. Neuroscience, 91(1), 391-400.
- Dubowy, C., & Sehgal, A. (2017). Circadian rhythms and sleep in Drosophila melanogaster. Genetics, 205(4), 1373-1397.
- Shafer, O. T., & Keene, A. C. (2021). The regulation of Drosophila sleep. Current Biology, 31(1), R38-R49.
- Allada, R., & Siegel, J. M. (2008). Unearthing the phylogenetic roots of sleep. Current biology, 18(15), R670-R679.
- Siegel, J. M. (2005). Clues to the functions of mammalian sleep. Nature, 437(7063), 1264-1271.
- Diekelmann, S., & Born, J. (2010). The memory function of sleep. Nature Reviews Neuroscience, 11(2), 114-126.
- Tononi, G., & Cirelli, C. (2014). Sleep and the price of plasticity: from synaptic and cellular homeostasis to memory consolidation and integration. Neuron, 81(1), 12-34.
- Tononi, G., & Cirelli, C. (2016). Sleep and synaptic down-selection. Micro-, meso-and macro-dynamics of the brain, 99-106.
- Xie, L., Kang, H., Xu, Q., Chen, M. J., Liao, Y., Thiyagarajan, M., … & Nedergaard, M. (2013). Sleep drives metabolite clearance from the adult brain. science, 342(6156), 373-377.
- Van Alphen, B., Semenza, E. R., Yap, M., Van Swinderen, B., & Allada, R. (2021). A deep sleep stage in Drosophila with a functional role in waste clearance. Science advances, 7(4), eabc2999.
- Borbély, A. A., Daan, S., Wirz‐Justice, A., & Deboer, T. (2016). The two‐process model of sleep regulation: a reappraisal. Journal of sleep research, 25(2), 131-143.
- Ko, C. H., & Takahashi, J. S. (2006). Molecular components of the mammalian circadian clock. Human molecular genetics, 15(suppl_2), R271-R277.
- Groos, G., & Hendriks, J. (1982). Circadian rhythms in electrical discharge of rat suprachiasmatic neurones recorded in vitro. Neuroscience letters, 34(3), 283-288.
- Takahashi, J. S., DeCoursey, P. J., Bauman, L., & Menaker, M. (1984). Spectral sensitivity of a novel photoreceptive system mediating entrainment of mammalian circadian rhythms. Nature, 308(5955), 186-188.
- Miyamoto, Y., & Sancar, A. (1998). Vitamin B2-based blue-light photoreceptors in the retinohypothalamic tract as the photoactive pigments for setting the circadian clock in mammals. Proceedings of the National Academy of Sciences, 95(11), 6097-6102.
- Edgar, D. M., Dement, W. C., & Fuller, C. A. (1993). Effect of SCN lesions on sleep in squirrel monkeys: evidence for opponent processes in sleep-wake regulation. Journal of Neuroscience, 13(3), 1065-1079.
- Stephan, F. K., & Zucker, I. (1972). Circadian rhythms in drinking behavior and locomotor activity of rats are eliminated by hypothalamic lesions. Proceedings of the National Academy of Sciences, 69(6), 1583-1586.
- Filipski, E., King, V. M., Li, X., Granda, T. G., Mormont, M. C., Liu, X., … & Lévi, F. (2002). Host circadian clock as a control point in tumor progression. Journal of the National Cancer Institute, 94(9), 690-697.
- Stephan, F. K. (2002). The “other” circadian system: food as a Zeitgeber. Journal of biological rhythms, 17(4), 284-292.
- Tahara, Y., & Shibata, S. (2018). Entrainment of the mouse circadian clock: effects of stress, exercise, and nutrition. Free Radical Biology and Medicine, 119, 129-138.
- Aschoff, J., Fatranska, M., Giedke, H., Doerr, P., Stamm, D., & Wisser, H. (1971). Human circadian rhythms in continuous darkness: entrainment by social cues. Science, 171(3967), 213-215.
- Golombek, D. A., Casiraghi, L. P., Agostino, P. V., Paladino, N., Duhart, J. M., Plano, S. A., & Chiesa, J. J. (2013). The times they’re a-changing: effects of circadian desynchronization on physiology and disease. Journal of Physiology-Paris, 107(4), 310-322.
- Challet, E. (2015). Keeping circadian time with hormones. Diabetes, Obesity and Metabolism, 17, 76-83.
- Gao, Q., Lv, J., Li, W., Zhang, P., Tao, J., & Xu, Z. (2016). Disrupting the circadian photo-period alters the release of follicle-stimulating hormone, luteinizing hormone, progesterone, and estradiol in maternal and fetal sheep. Journal of Reproduction and Development.
- Armario, A., Montero, J. L., & Jolin, T. (1987). Chronic food restriction and the circadian rhythms of pituitary-adrenal hormones, growth hormone and thyroid-stimulating hormone. Annals of nutrition and metabolism, 31(2), 81-87.
- Borb, A. A., & Achermann, P. (1999). Sleep homeostasis and models of sleep regulation. Journal of biological rhythms, 14(6), 559-570.
- Pappenheimer, J. R., Koski, G., Fencl, V., Karnovsky, M. L., & Krueger, J. (1975). Extraction of sleep-promoting factor S from cerebrospinal fluid and from brains of sleep-deprived animals. Journal of neurophysiology, 38(6), 1299-1311.
- Radulovacki, M. (1985). Role of adenosine in sleep in rats. Reviews in clinical & basic pharmacology, 5(3-4), 327-339.