Deprivation & Allostasis – The Cost of Sleep Loss and of Adaptation
February 20, 2022
ImmunoLP
September 3, 2022The Gut Microbiota – a Fundamental Mediator of Metabolism
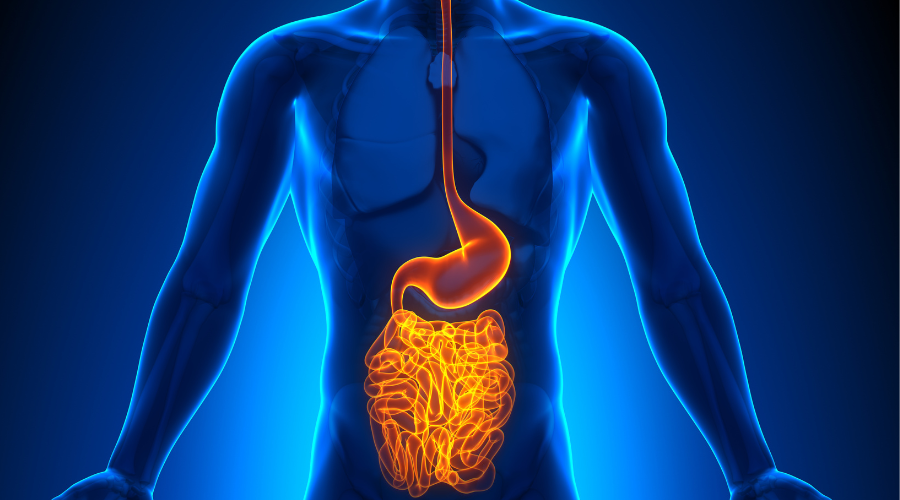
The complex ecosystem comprising trillions of microorganisms (bacteria, yeast, protozoa, and viruses) housed in the gut are known collectively as the gut microbiota. The collection of all gut microbial genes (the gut microbiome) are more than one order of magnitude higher than that of genes in the human genome. The majority of these microbes are commensal or mutualistic and reside in the digestive tract (especially the distal colon), but they are also present on our skin and elsewhere.Our co-existing microorganisms play a central role in human physiology. They are driven by the symbiotic exchange of metabolic products between host and microbe, and we are dependent on each other for numerous biological functions (e.g. nutrient absorption). It is now acknowledged that the gut microbiome plays a direct role in mediating communication between the different organ systems. There are clear links between the microbiome and its influence on host metabolism, with profound implications for human health given the prevalence of obesity and metabolic syndrome in Western societies.
The Interface between Diet, the Gut Microbiome, and Metabolic Health Part 1
Cutting edge techniques in microbiome research have revealed that environmental factors such as diet and lifestyle have more of an impact on shaping our gut microbiome than genetics.
Dietary fiber is a polymer of carbohydrates (oligosaccharides), and humans lack the enzymes to break down the glycosidic bonds. When we consume fiber, it passes through our digestive tract mostly undigested. When it reaches the colon, fiber gets metabolized through a process called fermentation whereby several carbon compounds are generated, principally the short chain fatty acids (SCFAs) butyrate, propionate, and acetate. Butyrate has been shown to be of particular importance. These SCFAs bind to receptors on various organ sites and elicit signaling cascades that promote a myriad of downstream effects. When SCFAs bind to receptors on the intestinal brush border, peptides such as peptide YY (PYY) and glucagon-like peptide-1 (GLP-1), which have appetite suppressing (anorexigenic) effects, are activated. Butyrate can bind to G-protein coupled receptors (GPCRs) on adipocyte (fat cell) membranes causing decreased fat accumulation, stimulation of lipolysis and beta-oxidation (so it promotes the release of fat from the fat cells and the oxidation of that fat). It also decreases insulin signaling, which further enhances fatty acid release and oxidation (colloquially known as “fat burning”). Insulin is a critical hormone, but too much is not good. Insulin inhibits lipolysis and beta-oxidation, causing fat accumulation. It also increases the transcription of enzymes involved in fat synthesis (lipogenesis) and decreases genes known to be involved in longevity.
Takeaway
Dietary fiber is the indigestible part of food-derived plant matter to which we lack the digestive enzymes to break down ourselves. Instead, the gut microbiome breaks it down and converts it into compounds such as SCFAs, which are cell signaling molecules that act on several different organ systems. SCFAs increase hormones that promote satiety such as PYY, GLP-1, and CCK. Butyrate in particular has many beneficial effects. It serves as a primary fuel source of our colon cells and for helping to maintain the integrity of the gut in many ways. It also stimulates fat loss and improves metabolic health through several mechanisms.
The Interface Between Diet, Gut Microbiome, and Metabolic Health Part 2
SCFAs can inhibit a protein inside enterocytes called Fiaf. Fiaf’s normal function is to inhibit lipoprotein lipase (LPL). So, the inhibition of Fiaf releases inhibition on LPL. This, in turn, promotes decreased fat accumulation. SCFAs also act on the adipose tissue to increase beta adrenergic signaling, which further increases fatty acid degradation to be released into the blood and oxidized. Additionally, SCFAs decrease adipose tissue inflammation and enhance the conversion of white adipose tissue (WAT) to brown adipose tissue (BAT), which has more of a propensity to burn fat and thermogenic effect. Furthermore, SCFAs also trigger the release of leptin from the adipose tissue, a hormone involved in satiety.
Further, these compounds also act on the liver, skeletal muscle, and colonocytes. Butyrate in particular serves as a fuel for colonocytes, reduces inflammation in the colon and helps to maintain the integrity of the tight junction structures as well as the pH in the lumen. It also promotes cell cycle arrest and apoptosis, thereby reducing colon polyps that could develop into colon cancer.
A paucity of SCFAs (by not getting enough through diet or by antibiotic depletion) contributes to disruption of microbial homeostasis. Pathogenic microbes, such as E. coli and Salmonella, are then given a more hospitable and opportunistic environment to outcompete commensal microbes that characterize a healthy gut flora. Microbial homeostasis is, in part, maintained by activation of receptors for SCFAs (such as PPAR-y).
Obesity and metabolic disorders such as type 2 diabetes are associated with lower microbial diversity and specific types of microbes. The capacity to harvest more calories from the diet is increased in obese individuals. Compared to lean mice and humans, obese individuals have an increased relative abundance of Firmicutes, and reduced abundance of Bacteroidetes. Metagenomic analyses demonstrate that individuals with reduced microbial diversity have higher levels of systemic inflammation, insulin resistance, dyslipidemia, and adiposity. Conversely, other genera including Akkermansia, Bifidobacterium, Lactobacillus, and Faecalibacterium have been demonstrated to be associated with higher microbial gene count and lean individuals with better overall metabolic health.
Takeaway
Obesity and metabolic disorders such as T2DM are associated with specific microbial signatures that correlate with insulin resistance, systemic inflammation, excess fat accumulation, and aberrant fat metabolism. Interestingly, when you swap the gut microbiome of two rodents–let’s say one that is obese with metabolic syndrome and one that is lean and metabolically healthy–it changes their phenotype to that of the one they were swapped for. So, the lean mouse becomes obese and insulin resistant while the obese mouse becomes lean and metabolically healthier. This suggests that the gut microbiome may have a direct causal role in our metabolic health.
The Influence of the Gut Microbiota on Metabolism Through Regulation of Gut Hormones
As highlighted previously, the gut microbiome contributes significantly to how we extract energy and nutrients from food by converting inaccessible dietary components (such as fiber and polyphenols) into an array of bioactive metabolites that are readily absorbed. These bioactive metabolites trigger signaling cascades that confer numerous downstream effects on host metabolism and can have long-term effects through epigenetic modifications.
The hormonal peptides mentioned earlier, GLP-1 and PYY, are synthesized and secreted within the mucosal lining of the gut by specialized enteroendocrine L cells. Additionally, these L cells also secrete other products involved in key metabolic processes such as cholecystokinin (CCK), serotonin (also known as 5-HT), and gastric inhibitory polypeptide (GIP). There is a bidirectional communication between the microbiome (and the metabolites they produce) in the lumen of the gut and the enteroendocrine cells.
A study by Lee et al showed that GLP-1 decreased inflammation in the fat tissue of diabetic mice. Decreasing inflammation in fat tissue improves insulin sensitivity and metabolic function. Just like people with diabetes and obesity suffer from impaired insulin signaling, they also often suffer from impaired GLP-1 signaling, which could be due to alterations in the gut microbiome.
The Gut Barrier Integrity and Metabolic Health
The gut barrier is a multi-layer functional and physical entity that has many important roles. It separates the luminal contents of the gut from the rest of the body and prevents things from migrating ectopically. When elements in our gut traverse the gut barrier and leak into the portal circulation, a condition known as metabolic endotoxemia occurs. Metabolic endotoxemia drives chronic inflammation which underlies metabolic dysfunction. The gut barrier also plays an important role in the absorption of nutrients, electrolytes, and water.
A layer of mucus composed of complex glycoproteins, secreted by goblet cells, covers the inside of the gut epithelium to coat and protect it from invading pathogens and digestive enzymes. The mucus is infused with antibodies and antimicrobial agents to further protect the gut lining. It also serves as a lubricant and vehicle between luminal contents and intestinal epithelium. A thinner mucosal layer with fewer antimicrobial agents is observed in germ-free mice devoid of a gut microbiome, and is associated with metabolic dysfunction.
Part of the gut barrier contains a selectively permeable membrane composed of epithelial cells that are bound together by tight junction proteins which only allow desired nutrients to get through. When the integrity of the tight junction proteins are disrupted, harmful substances (pathogens and toxins) are able to enter. The tight junctions are heavily influenced by the gut microbiome and the substrates it is exposed to. This dynamic architecture can be revived by certain nutritional interventions. Some vitamins show protective effects on intestinal permeability. For example, vitamin D upregulates the expression of the tight junctions proteins zonulin-1, claudin-1, and E-cadherin.
A compromised gut barrier can be detected by measuring levels of LPS in the blood, which is associated with insulin resistance, obesity, and diabetes. High levels of dietary fructose have been shown to disrupt gut barrier integrity and enable the translocation of LPS. Mechanistically, it has been shown to do this by reducing mucus thickness, reducing expression of antimicrobial proteins, and downregulating tight junction proteins. A link between fructose consumption and the development of poor metabolic health outcomes has been demonstrated in several lines of evidence. Nutritional interventions such as probiotics, prebiotics, and various dietary sources have been shown to reduce the level of LPS in the blood as well as markers of inflammation in people with obesity, diabetes, and metabolic dysfunction.
For further reading on how various dietary compounds influence intestinal barrier integrity, click here and here
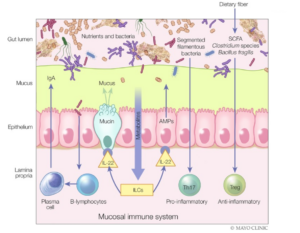
Photo credit: Camilleri et al., 2019
Takeaway
The gut barrier is a highly complex, multi-layer entity that plays many critical roles in keeping everything in check. It serves as an interface between the inner components of our gut and the rest of the body, preventing things from going where they do not belong. You may have heard the concept of a “leaky gut” before, which is the colloquial terminology for “intestinal permeability.” This refers to when the gut barrier is disrupted, causing many things to go awry. Substances that do not belong in the portal circulation are able to slip through the tight junctions and cross over to wreak havoc. For example, LPS, which is a membrane component of gram-negative bacteria, gets into the circulation and can cause “metabolic endotoxemia” which is a phenomenon that is highly toxic to the body and is linked to metabolic disorders. The gut microbiome plays an important role in maintaining the integrity of our gut barrier. Our environment, especially our diet, significantly influences our gut microbiome, which influences the integrity of our gut barrier.
Summary
Emerging data has illuminated the role of gut dysbiosis in the etiology of and pathogenesis of metabolic disorders including metabolic syndrome, type 2 diabetes, obesity, and non-alcoholic fatty liver disease. These metabolic disorders share underlying patterns of gut microbiome perturbation and intestinal barrier dysfunction. However, there is still much debate on the precise microbial signature of a healthy or dysbiotic gut microbiome. An individual’s genes can influence the gut microbiome’s composition and in turn can shape the person’s phenotype. However, our environment has a significant impact on our gut microbiome, so despite our genetic blueprint and mode of delivery at birth, as well as other factors we had no control over (whether we were bottle fed or breast fed, for example), the malleability of the gut microbiome gives us agency over our gut health, which in turn has significant influence on our metabolism and many other aspects of health not discussed here. Clearly, the gut microbiome serves as a critical mediator for many physiological and metabolic functions. Thus, an aberrant gut microbiome would have many adverse clinical consequences. Judicious use of probiotics, prebiotics, and various dietary constituents can have favorable effects on metabolic health mediated by the microbiome.
Thousands of studies and years of research have led us to create the most potent & clinically validated formula on the market to help normalize and regulate metabolic, immune, and gut health functions. Shop HARMONY, your protection against an imperfect lifestyle.