Part 1 How Ultraviolet Radiation Damages the Skin and Causes Photoaging
October 24, 2022
Part 3 Strategies to Combat Aging
December 11, 2022Part 2 The Impact of Advanced Glycation End Products on Collagen
Collagen is the most abundant structural protein in the body that makes up our skin, cartilage, bone, blood vessels, and connective tissue. Elastin is another protein in the extracellular matrix (ECM) that provides elasticity to our skin and connective tissues. Together, they play fundamental roles in the structure, resilience, elasticity, function, and biomechanical properties of an array of organs and tissues. The physiology of connective tissue is highly dependent on collagen and other ECM proteins that are hierarchically organized within the tissues. Collagen is one of the strongest proteins. In the skin, it not only serves as a supportive framework for cells and tissues, but also interacts with other cells and affects various cellular functions such as migration, differentiation and proliferation.
There are 28 known types of collagen which vary in the type of structures they form. The most common types are I, II, II, IV, and V. Type I is the most abundant, accounting for over 90% in humans.
Collagen is composed of the amino acids glycine, proline, and hydroxyproline. These amino acids form three strands, which make up the triple-helix structure characteristic of collagen. Since glycine is the smallest of all the amino acids, it allows the chain to form a tight configuration, providing some of its resilience.
One of the hallmarks of aging is disruption to the integrity of collagen. This goes for both skin aging and global aging of the organism.
What are Advanced Glycation End Products?
Advanced glycation end products (AGEs) are a diverse group of heterogeneous compounds formed by a series of non-enzymatic reactions between reducing sugars (or their derivatives) and the free amino groups of proteins, nucleic acids, or aminophospholipids. There are three different modes of AGE formation: Maillard reaction, the polyol pathway, and lipid peroxidation. The Maillard reaction is the browning you see on cooked foods and baked goods. AGEs are ubiquitous and we can be exposed to them exogenously (diet, cigarette smoke, pollution, ultraviolet radiation, other environmental elements) or through endogenous production (produced within the body, hyperglycemic conditions, normal aging and metabolism, inflammatory conditions, metabolic diseases). They are one of the major drivers of aging.
More than 20 different AGEs have been identified in human blood and tissues and in foods. The most prominent ones include carboxymethyl-lysine, carboxyethyl-lysine, pyrraline (nonfluorescent AGEs), pentosidine, and methylglyoxal-lysine dimer.
Below is a schematic depicting different types of AGEs.
What are Advanced Glycation End Products?
Advanced glycation end products (AGEs) are a diverse group of heterogeneous compounds formed by a series of non-enzymatic reactions between reducing sugars (or their derivatives) and the free amino groups of proteins, nucleic acids, or aminophospholipids. There are three different modes of AGE formation: Maillard reaction, the polyol pathway, and lipid peroxidation. The Maillard reaction is the browning you see on cooked foods and baked goods. AGEs are ubiquitous and we can be exposed to them exogenously (diet, cigarette smoke, pollution, ultraviolet radiation, other environmental elements) or through endogenous production (produced within the body, hyperglycemic conditions, normal aging and metabolism, inflammatory conditions, metabolic diseases). They are one of the major drivers of aging.
More than 20 different AGEs have been identified in human blood and tissues and in foods. The most prominent ones include carboxymethyl-lysine, carboxyethyl-lysine, pyrraline (nonfluorescent AGEs), pentosidine, and methylglyoxal-lysine dimer.
Below is a schematic depicting different types of AGEs
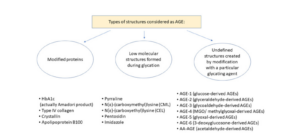
Photo credit: Kuzan, A., 2021
How AGEs Are Formed
Dietary AGEs are mainly formed by high heat cooking and food processing techniques (especially dry-heat) such as frying, baking, grilling, carmelizing, searing, and barbecuing. While cooking is known to increase AGE formation in foods, even uncooked foods can contain large quantities of AGEs. This is probably due pasteurization, curing, or aging (as in aged cheeses). For example, many animal-derived foods contain high levels of AGEs, such as higher fat aged cheese.
The generation of AGEs via the Maillard reaction occurs in three phases. First, a sugar molecule non-enzymatically attaches to a free amino acid from a protein, lipid, or nucleic acid forming a Schiff base. During the second phase, the Schiff base undergoes a conformational change and forms early glycation products, known as Amadori products. The reaction is still reversible at this point. Further crosslinking and chemical rearrangement occurs with these molecules, ultimately becoming AGEs.
Higher pH conditions can also increase the formation of AGEs, because the alkalinity promotes amino groups being in the basic deprotonated form, which increases reactivity.
The polyol pathway is another well studied mechanism by which AGEs form. This is when glucose is converted to sorbitol via aldose reductase and then to fructose via another enzymatic reaction. Fructose metabolites transform into aldehydes and interact with monoacids to become AGEs. Finally, the oxidation of glucose or peroxidation of lipids are other pathways by which AGEs form. Given the different ways AGEs can form, this may partly explain their heterogeneity.
Below is a simplified schematic illustrating how AGEs are formed.
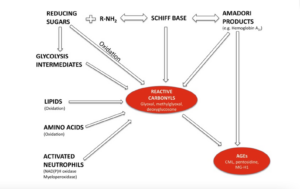 Photo credit: Uribari et al., 2015
Photo credit: Uribari et al., 2015
The Deleterious Effects of AGEs
AGEs induce pathology by two main mechanisms: 1) They directly crosslink with proteins, impairing their structure and function. 2) They activate intracellular signaling pathways through both receptor-mediated and non-receptor mediated mechanisms, leading to an increase in pro-inflammatory cytokines and oxidative stress. One such example is RAGE, which is one of the best studied receptors for AGEs.
AGEs play a role in the pathophysiology of many different diseases, including type 2 diabetes mellitus (T2DM, Alzheimer’s disease (AD), kidney failure, atherosclerosis, etc.
The microvascular and macrovascular damage seen in diseases such as diabetes is attributed to the accumulation of AGEs in tissues. AGEs cause stiffening and loss of elasticity of the arteries as well.
How AGEs are Destructive to Collagen
ECM proteins have been regarded as one of the major target structures for glycation. AGEs easily accumulate in the extracellular matrix of the dermis, which is where longevity proteins such as collagen, fibronectin, and elastin are. AGEs crosslink to these proteins and cause damage to their structure and biological properties. Collagen is particularly susceptible to destruction by AGEs due to the irreversibility of non-enzymatic cross-linking and the low turnover rate of collagen. Also, the levels of AGEs in collagen increase linearly with age. The proteins in the extracellular matrix, such as collagen, are destroyed with long term accumulation of AGEs, and the fibers become deformed, making them unable to maintain biomechanical properties. The appearance of glycated collagen is first observed at the age of 20. It accumulates with a yearly rate of about 3.7% reaching a 30–50% increase at 80 years old.
AGEs damage collagen in various ways. Intermolecular crosslinks of adjacent collagen fibers alter its biomechanical properties, leading to weakness, stiffness and reduced flexibility. The formation of AGEs on the side chains of collagen and alteration of its charge inhibits its ability to interact with its contact sites and other ECM proteins.
When collagen is impaired in this way, it is resistant to being eliminated and replaced by newly synthesized, functional collagen. Also, AGEs are very reactive molecules. As early as during their crosslinking reactions they can act as electron donors leading to formation of superoxide anions and other reactive oxygen species. The glycation of proteins creates cation-radical sites in the proteins that are able to catalyze oxidation-reduction reactions leading to ROS generation with or without presence of oxygen or transition metals such as iron and copper.
Multiple studies have demonstrated that AGEs have a strong impact on skin physiology and are a pathogenic factor for skin complications of metabolic diseases such as impaired wound healing, diabetic skin ulcers, and infections. When AGEs increase in the skin, participants of these studies acquired various skin problems such as deepening of wrinkles, poor elasticity, apoptosis of skin-related cells, inflammation, and pigment issues (yellowing and browning).
Ultraviolet Radiation and Collagen
As mentioned in article one of this series, another major factor that breaks down collagen is ultraviolet radiation (UVR), especially UVA rays, which are the predominant UV ray in the atmosphere. UVA rays penetrate deep into the dermal layers of the skin and damage collagen fibers. UVR upregulates collagen-cleaving matrix metalloproteins (MMPs) which break down collagen and inhibit pro-collagen synthesis. This causes premature wrinkles, sagging, and global skin aging. UVR also promotes the formation of AGEs.
Summary
UVR and AGEs are two major drivers of skin aging. AGEs are a cluster of heterogeneous molecules that are formed through nonenzymatic glycation of proteins, lipids, or nucleic acids and subsequent chemical rearrangements. They accumulate in all tissues and can cross-link with proteins, especially collagen due to its slow turnover rate and high binding affinity for reducing sugars. AGEs are difficult to eliminate from the body and are irreversible once they are formed. This cross-linking with other proteins, such as collagen, damages them and impairs their functionality. Furthermore, AGEs can act as a ligand to modulate signaling pathways that promote inflammation and excess reactive oxygen species.
Some of the damage AGEs confer to their target proteins, lipids, and nucleic acids are modification of DNA regulation and epigenetic modulation, enzyme-substrate interactions, protein-DNA interactions, protein-protein interactions. Therefore, AGEs interfere with many physiological functions.
AGEs have a substantially wide-reaching impact. Not only can they react with proteins, lipids, and nucleic acids in virtually all tissues and skin cells, but they can also affect intracellular and extracellular biomolecules. Through alteration of the physicochemical properties, increased cellular senescence, upregulation of oxidative stress and inflammation, AGEs significantly contribute to the overall picture of skin aging. Evidence regarding the pivotal role that AGEs play in aging has emerged over the years, and potential strategies to combat AGEs have received high levels of interest. Article 3 of this series will delve into the evidence for ways to combat these major drivers of aging (UVR and AGEs, in particular).